
In previous articles we have been seeing what the Kinetic energy and mechanical energy. In these articles we mentioned thermal energy as part of the energy that influences and possesses the body in question. Thermal energy It is the energy that all the particles that make up a body have. When the temperature oscillates between increase and decrease, the activity of the body increases. This internal energy increases as the temperature is higher and decreases when it is lower.
Now we are going to thoroughly analyze this type of energy and further complete our knowledge about the different types of energy that exist. Do you want to learn more about it? Keep reading and you will find out.
Characteristics of thermal energy

It is the energy that interferes in the different calorific processes that occur when bodies of different temperatures come into contact. As long as the bodies maintain a friction between them, this energy will be transmitted from one body to another. This is what happens, for example, when we place our hand on a surface. After a while, the surface will have the temperature of the hand, because he has given it to him.
The gain or loss of this internal energy during the process it is called heat. Thermal energy is acquired from a number of different means. Therefore, every body that has a certain temperature has an internal energy inside.
Examples of thermal energy
Let's take a closer look at the sources for acquiring thermal energy:
- Nature and the Sun They are two sources of energy that provide internal energy to bodies. For example, when an iron is continuously exposed to the sun, its temperature rises because it absorbs internal energy. In addition, the star king is the clearest example of thermal energy. It is the largest known source of thermal energy. Animals that are unable to regulate their temperature take advantage of this source of energy to do so.
- Boil water: As the water temperature increases, the thermal energy of the entire system begins to multiply. The time came when the increase in temperature in thermal energy forces the water to a phase change.
- Fireplaces: the energy produced in the chimneys comes from the increase in thermal energy. Here a combustion of organic matter is maintained so that the home can be kept warm.
- Heater: serves to increase the temperature of water in a similar way to when we are boiling.
- Exothermic reactions that occur through the burning of some fuel.
- Nuclear reactions that take place by nuclear fission. It also occurs when it occurs by fusion of the nucleus. When two atoms have a similar charge, they join together to have a heavier nucleus and during the process they release a large amount of energy.
- The joule effect occurs when a conductor circulates an electric current and the kinetic energy that electrons have are transformed into internal energy as a result of continuous collisions.
- Friction force It also generates internal energy, since there is also an energy exchange between two bodies, whether it is a physical or chemical process.
How is thermal energy produced?
We have to think that energy is neither created nor destroyed, but only transformed. Thermal energy is generated in many ways. It is generated by the movement of atoms and molecules of matter like a form of kinetic energy that is produced by random movements. When a system has a greater amount of thermal energy, its atoms move faster.
How is thermal energy used?
Thermal energy can be transformed by a heat engine or mechanical work. Among the most common examples is the engine of a car, plane or boat. Thermal energy can be harnessed in many ways. Let's see which are the main ones:
- In those places where heat is needed. For example, as heating in a home.
- Conversion of mechanical energy. An example of this are the combustion engines in cars.
- Electrical energy transformation. This is generated in thermal power plants.
Internal energy measurement
Internal energy is measured according to International System of Units in Joules (J). It can also be expressed in calories (Cal) or kilocalories (Kcal). To understand internal energy well, we must remember the principle of conservation of energy. "Energy is neither created nor destroyed, it only transforms from one to the other." This means that even though the energy is continually transforming, it is always the same amount.
The kinetic energy that a car carries when it hits a building goes directly to the wall. Therefore, as a result, its internal energy increases and the car decreases its kinetic energy.
Examples of thermal energy
The heat or thermal energy is for example in:
- Warm-blooded animals. For example, when we feel cold we hug others. So little by little we feel better, since it transfers its heat to us.
- On the metal exposed to the sun. In summer especially it gets to burn.
- When we put an ice cube in a cup of hot water we see that it melts because the heat is conducted to it.
- Stoves, radiators, and in any other heating system.
Frequent confusion
It is very common to confuse heat energy with thermal energy. It is often widely used as synonyms even though they have nothing to do with it. Heat energy focuses exclusively on the emanation of heat in its caloric phenomena. Therefore, it is distinguished from thermal energy which is only heat.
The amount of heat in a body is the measure of thermal energy, while the heat that can emanate from the body indicates that it has a higher thermal energy capacity. The temperature of a body gives us the sensation of heat and can give us a signal that indicates the amount of thermal energy it has. As we said before, the more temperature the body has, the more energy.
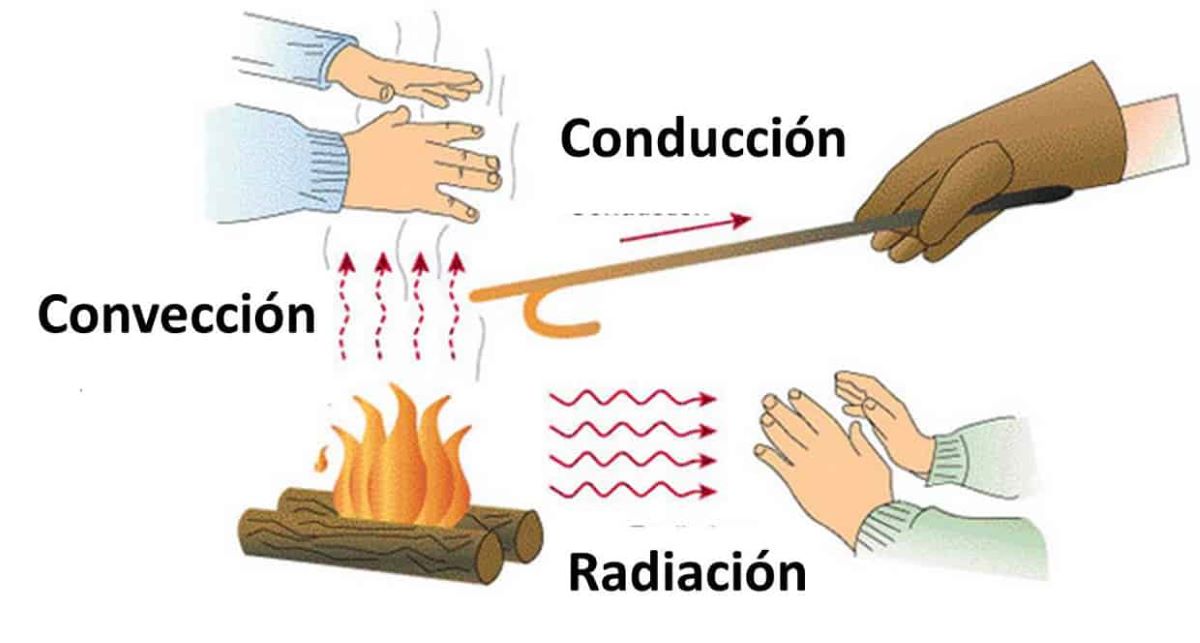
Heat can be transmitted in many different ways. Let's review them one by one:
- Electromagnetic wave radiation.
- Driving. When energy is transmitted from a warmer body to a cooler body, conduction occurs. If the bodies are at the same temperature, there is no energy exchange. The fact that the two bodies equal their temperature when they are in contact is another principle of physics called thermal equilibrium. For example, when we touch a cold object with the hand, the thermal energy is transmitted to the object causing the sensation of cold in our hand.
- Convection. This occurs when the hottest molecules are transformed from one side to the other. It takes place in nature continuously in the wind. The hottest particles tend to move where there is less density.
Other related energies
Thermal energy is related to many other forms of energy. Here we have some of them.
Solar thermal

It is a type of renewable energy that consists of the transformation of solar energy into heat. This energy is used to heat water for different uses such as domestic or in hospitals. It also serves as heating on winter days. The source is the sun and it is received directly.
Geothermal Energy
Obtaining thermal energy causes an environmental impact due to to the release of carbon dioxide and radioactive waste. However, if energy from the interior of the earth is used. It is also a type of renewable energy that does not pollute or cause damage to the environment.
Electrical and chemical energy
Thermal energy can be transformed into electrical energy. For example, fossil fuels generate electricity by burning and releasing it. Electric energy is given as a result of a potential difference between two points and allows to create an electric current between both when they come into contact with an electrical conductor. The conductor can be a metal.
Thermal energy is a type of energy released in the form of heat due to the contact of a body with a higher temperature to another with a lower temperature, as well as it can be obtained by different situations or means as previously mentioned. Chemical energy is the one that has a chemical bond, that is to say, it is an energy produced solely by chemical reactions.
With this information you will be able to better understand thermal energy.